DSHEA Tutorial: Ingredient Listing

This is another rather easy part, it’s simply a list of your ingredients.
This list of ingredients is supposed to be found on the panel to the right of your primary display panel (from the perspective of the consumer looking at the label.) This panel is called the “information panel”.
- The list must be preceded by the word “Ingredients”. Is that so difficult?
The use of the word "Ingredients" on a herbal supplement label
- The size of the type can be no smaller than 1/16 inch when measuring the height of the lower case “o”. Again, you’ll need to eventually print this out to assess the size of the lower case letter “o”. Don’t use anything that your computer says or measure it onscreen.
- The ingredients must be listed in descending order of weight. That means that the herb that is the greatest weight in the formula gets listed first as opposed to the names of the king herbs (most important herbs in the formula, in the case of Shen Ling Bai Zhu San they would be Ren Shen, Fu Ling, and Bai Zhu). It is common knowledge that many extract powders, especially those from Taiwan also include fillers to ensure consistent potency between batches. I have yet to see these fillers listed on any of the bottles that contain these Taiwanese extract powders. As such, I have not included fillers on my bottle label ingredients. It would be prudent to look into this further sometime in the future. This issue applies to the capsules you’ll use for your product too. I have seen in the FDA’s examples of labels (see prior page) that the ingredient of “gelatin” is used to describe the capsule. You’ll probably want to add that.
So, now we come to an issue that doesn’t actually show up in the Dietary Supplement Labeling Guide. In fact there is some controversy regarding this next point, but I’ll pass on what I’ve figured out.
What DSHEA really wants from us is to list the “common name” plus the Latin binomial including the part of the plant. Now there are a few herbs where the latin in one book doesn’t really agree with the latin from another. So which do you chose? It appears that the FDA is defaulting to the list of herbs that is available through one of two herbal medicine books.
According to the FDA website, the current regulation (Aug. 2003) incorporates the references available in Herbs of Commerce
and the International Code of Botanical Nomenclature (Tokyo Code) 2000.
If you don’t own those two books, you’re probably okay with using one of the better Chinese herbal textbooks available such as either of the Eastland Press books on herbs and/or formulas, or the Chen & Chen texts on Chinese herbs and formulas.
When it comes to the “common” term that the FDA suggests, I’m guessing that they’re asking for the English language common term. However many Chinese herbs do not have an English equivalent, or the English term is a modernized version of the Latin, or the common English term is so unknown as to render them somewhat, dare I say, uncommon. Certainly, within the TCM industry we’re more commonly using terms such as “dang gui” than “angelica”. So for my labels, I’m using the Pinyin plus the Latin binomial with plant part. “Binomial” means “two names” so, be sure and include both of those longish Latin words including the part of the plant or animal used.
Here’s an example:
Dang Gui – Rx. Angelicae Sinensis
I guess you could make a case for “Dong Quai” being the “common” term, but that’s the old Wide-Giles transliteration scheme which is not as commonly used anymore, so I’ll favor the Pinyin instead.
Another example:
Fu Ling – Scl. Poria Cocos
Because Fu Ling is such an unknown entity in the English speaking world, we’re limited to these bizarre “common names” such as “sclerotum of tuckahoe”, “china-root”, “poria”, “hoelen”, and my personal favorite: “Indian bread”. This is why I prefer to default back to the Pinyin which at least TCM practitioners use, but that’s not the official party line, just my opinion for your information.
So, now let’s add the ingredients to that panel to the right of the principal display panel. This panel to the right is called the “information panel”.
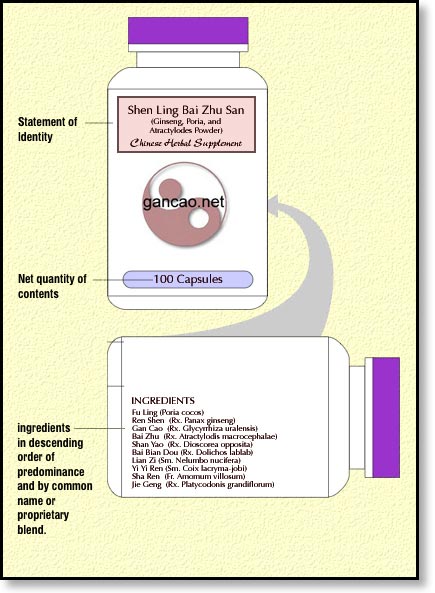
Ingredients list according to DSHEA guidelines.
Next: who’d you say you were?
 Last modified: August 26, 2009
Last modified: August 26, 2009  Tags: DSHEA, Labeling · Posted in: Labeling
Tags: DSHEA, Labeling · Posted in: Labeling
