DSHEA Tutorial: Dosage, Directions For Use

Next we have the so-called Directions for use (e.g., “Take five capsules, three times daily.”).
On some of the FDA labeling guidelines, this is required, on others it is not. However by including this information you not only help the consumer derive a successful response from the herbs, but you can also protect yourself from liability should the consumer misuse the product.
American consumers are accustomed to the “One-A-Day” approach to taking things that aren’t drugs. Even five capsules, three times per day is a lot of capsules for someone who is used to taking two Contact capsules every eight hours. So it’s best to make sure that the consumer knows how much to take.
By putting that suggested dosage on the label, you also protect yourself from someone taking the entire bottle in one or two sittings. In fact, one of the defense arguments for the (recently reversed) reversal of the ban on Ma Huang Ephedra was that the labeling of the bottle in question gave suggested dosages which were well within safe quantities. By “(recently reversed) reversal”, I mean that first the FDA said we couldn’t sell ephedra in dietary supplements. That was reversed because the suggested dosage was not unsafe, but a higher court reversed that decision and so ephedra is currently unavailable. The only court left is the US Supreme Court. Not sure if the dietary supplement industry is able to go that far. Stay tuned. (and check the date on the bottom of this page to assess how up-to-date this statement is.)
Let’s go ahead and add that dosage to the labels now.
The “suggested use” message can go on the same face as the ingredients. If you would prefer, you can put it on the primary panel too. If there is no room on either the primary or information panels, you can keep moving to the right (while facing the bottle). [source]
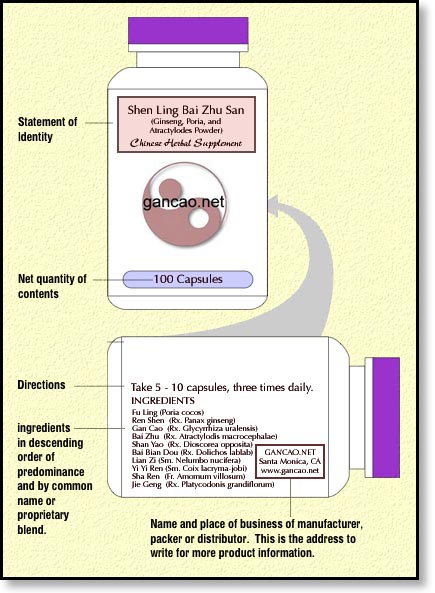
Suggested Use on a DSHEA label
Next: review of labeling requirements.
 Last modified: August 24, 2009
Last modified: August 24, 2009  Tags: DSHEA, Labeling · Posted in: Labeling
Tags: DSHEA, Labeling · Posted in: Labeling
